By Waseem Khan
A clinical trial coordinator in a U.S. oncology centre spends more time reconciling spreadsheets than reviewing patient outcomes.
Data flows in continuously-from imaging systems, laboratory platforms, electronic health records, and external research partners. Yet despite the volume, very little of it arrives in a form that can be trusted, aligned, or immediately reused. Teams compensate through manual workarounds: emails, duplicated files, disconnected trackers, and repeated validation cycles.
In a field where time directly influences patient outcomes, the issue is not a lack of data.
It is the absence of a system capable of turning that data into usable intelligence.
The Hidden Infrastructure Problem in Oncology Research
Modern oncology has become one of the most data-intensive domains in healthcare. Precision medicine, genomic analysis, and multi-centre clinical trials all depend on the ability to manage, share, and interpret complex datasets at scale.
However, much of the underlying infrastructure has not evolved at the same pace.
Research environments remain fragmented across:
Electronic Health Records (EHRs)
Laboratory and imaging systems
Document repositories
Compliance and audit platforms
These systems are rarely designed to operate as a unified whole. As a result, interoperability becomes a persistent challenge, particularly in multi-institutional trials where coordination is critical.
The consequences are systemic rather than technical. Clinical trials are delayed. Data inconsistencies emerge. Administrative overhead increases. Most importantly, researchers lose time-time that is often irreversible in a clinical context.
This is not a tooling issue. It is an architectural one.
From Systems to Intelligence
What is increasingly required is not simply better software, but a different way of designing research environments altogether.
Oncology research does not suffer from a lack of systems. It suffers from a lack of integrated, governed intelligence.
This distinction matters.
Traditional IT approaches focus on implementing platforms. Intelligent infrastructure focuses on how data, workflows, compliance, and collaboration operate together as a single, coherent system.
The shift is from:
Systems → to structured environments
Data → to governed information
Processes → to automated workflows
Outputs → to actionable insight
This is where the IntelliCare framework emerges-not as a product, but as a structured architectural approach to modernising oncology informatics.
The IntelliCare Framework: Engineering Research Environments for Scale
The IntelliCare framework represents a move toward intelligent, governed, and automated research ecosystems. Rather than layering new tools onto existing fragmentation, it introduces a unified architecture designed to support scalability, compliance, and high-velocity research.
Figure 2: The IntelliCare Framework – Intelligent Oncology Research Architecture
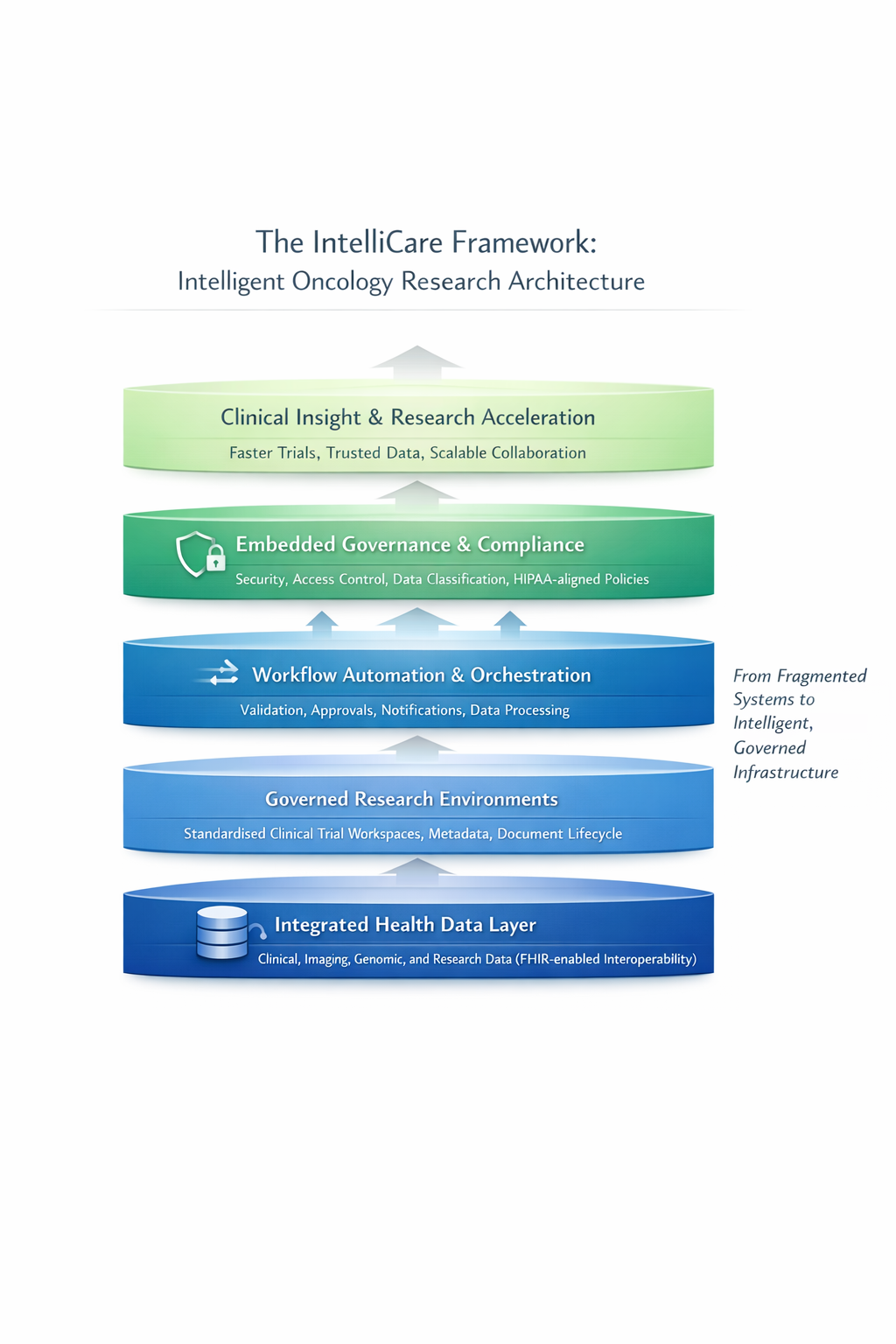
At its core, the framework integrates four critical dimensions.
- Integrated Health Data Architecture
Clinical, imaging, and research data are brought together through interoperable standards such as FHIR, enabling structured and consistent data exchange across systems. This removes the need for manual transformation and reconciliation.
- Governed Research Workspaces
Each clinical trial or research initiative is provisioned within a controlled digital environment, with standardised templates, metadata structures, and document lifecycle management. This ensures consistency, traceability, and audit readiness from the outset.
- Workflow Automation
Manual processes-approvals, notifications, compliance checks, and data validation-are replaced with automated workflows. For example, patient enrollment can trigger a sequence of compliance verification, documentation, and stakeholder communication without human intervention.
- Embedded Data Governance and Compliance
Security and compliance are not applied after the fact; they are built into the architecture. Data classification, retention policies, and access controls are enforced automatically, ensuring alignment with regulatory requirements such as HIPAA.
The result is not simply operational efficiency, but a fundamental shift in how research environments function. Administrative friction is reduced, data becomes more reliable, and collaboration becomes scalable.

Figure 1: From Fragmented Oncology Systems to Intelligent Research Infrastructure
Resolving the Constraints That Slow Research
One of the less visible challenges in oncology infrastructure lies in the underlying architectural constraints that prevent systems from scaling effectively.
For example, issues such as conflicting schema definitions within SharePoint-based environments-often dismissed as minor technical problems-can prevent the automated provisioning of research workspaces. In practice, this delays the setup of clinical trials and introduces manual overhead into processes that should be standardised.
Addressing these constraints requires more than configuration. It requires architectural intervention.
By developing automated remediation approaches-such as identifying and resolving conflicting data structures at the system level-it becomes possible to enable the rapid deployment of consistent, governed research environments. This allows institutions to scale from managing a handful of trials to supporting hundreds concurrently.
In high-volume oncology research, this difference is not incremental. It is transformational.
Security and Compliance as Architectural Foundations
Oncology data is among the most sensitive in healthcare, encompassing patient identities, treatment histories, and genomic information. Traditional approaches often treat security as a reactive layer, applied after systems are implemented.
This is no longer sufficient.
A resilient research environment requires a proactive model where security and compliance are embedded into the architecture itself.
This includes:
Role-based access controls that limit data exposure to what is necessary
Automated classification of sensitive data
Data loss prevention mechanisms to prevent unauthorised sharing
Continuous audit and monitoring for regulatory reporting
For instance, if an attempt is made to export patient-identifiable data without appropriate permissions, the system should not only block the action but also record and report it automatically.
This approach ensures that research can scale without increasing risk-a critical requirement in regulated healthcare environments.
Enabling the Workforce, Not Just the Technology
A common bottleneck in healthcare transformation is the reliance on specialised IT teams to design and maintain complex systems. This creates delays, limits innovation, and restricts scalability.
The IntelliCare approach addresses this by extending capability beyond IT.
Through the use of low-code platforms and governed development environments, clinicians, researchers, and operational staff can participate directly in building and refining workflows. This reduces dependency on central teams while maintaining oversight and control.
The impact is twofold:
Faster deployment of solutions
Increased digital capability across the organisation
In effect, the workforce becomes an active participant in the evolution of the system, rather than a passive user of it.
From Institutional Efficiency to National Impact
While the challenges of oncology infrastructure are often discussed at the institutional level, their implications extend much further.
At scale, the ability to manage and mobilise research data effectively contributes to broader national outcomes.
Accelerated clinical trials lead to faster drug discovery and approval. Improved data accessibility enables better analysis of treatment effectiveness and population health trends. Reduced administrative overhead allows resources to be redirected toward patient care and innovation.
Perhaps most importantly, standardised and interoperable systems enable collaboration across institutions-strengthening the overall research ecosystem.
In this context, oncology informatics is not simply a technical domain. It is a component of national healthcare capability.
The Shift Ahead
Oncology research is entering a phase where the volume and complexity of data will continue to increase exponentially. Traditional systems, built for a different era, are no longer sufficient to support this reality.
The future will not be defined by organisations that generate the most data.
It will be defined by those that can structure, govern, and apply that data as intelligence.
Frameworks such as IntelliCare represent an early step in this direction-moving from fragmented systems toward integrated, intelligent infrastructure capable of supporting both clinical advancement and long-term resilience.
Because in oncology, as in all high-stakes environments, the true advantage does not come from information alone.
It comes from the ability to use it-consistently, securely, and at speed.
About the Author
Waseem Khan is a cloud architecture and digital transformation specialist with over 13 years of experience delivering enterprise-scale solutions across healthcare, government, and regulated sectors. He works with senior leaders to design and implement intelligent, secure, and scalable digital environments that align data, workflows, and compliance with accountable governance and operational performance. His work focuses on modernising clinical and research systems, particularly in oncology, through structured architectural frameworks that integrate automation, interoperability, and data governance. Check his LinkedIn.